Super fact 97 : Things that spin have an angular momentum and if electrically charged they also generate a magnetic field (magnetic dipole). This is true for electrons and many other elementary particles. However, electrons are not physically spinning. This “spin” is an intrinsic property like electrical charge or mass. In addition, the electron has “Spin 1/2,” meaning you must rotate its wave function 720 degrees (two full circles) to get back to where you started. Moreover, this half spin oddity makes the existence of matter possible.
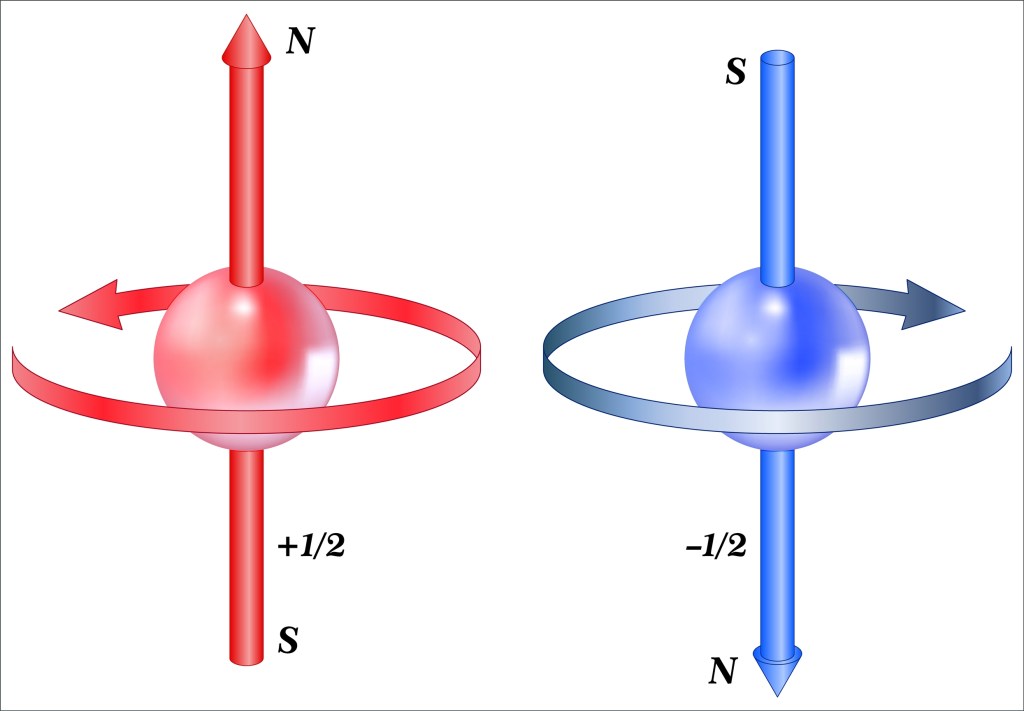
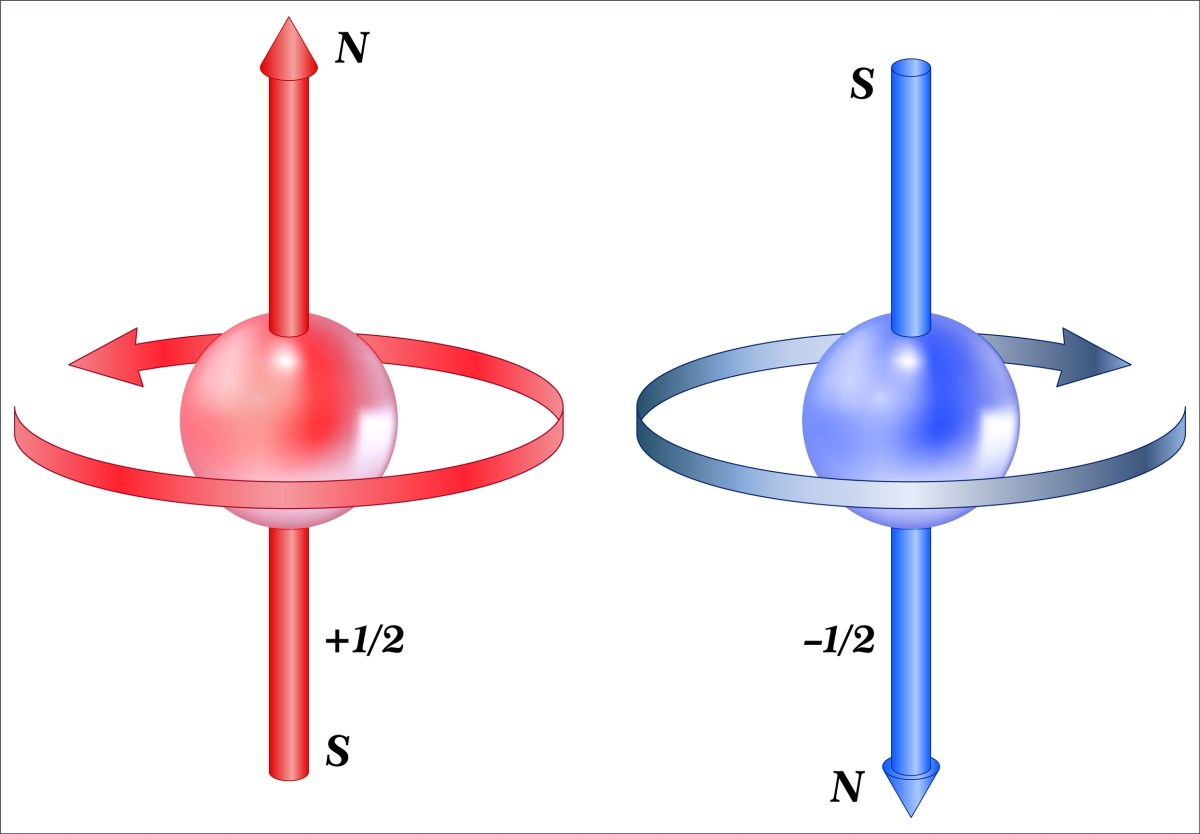
An electron with spin +1/2 will align parallel with an external magnetic field while an electron with spin -1/2 will align in the opposite direction. The existence of intrinsic spin and the fact that the half spin property requires you to rotate the electron’s wave function twice to get back to where you started is very surprising. Particles with half spin (electrons, protons, positrons, muons, etc.) are called Fermions. It is also surprising that the half spin property makes the existence of matter possible. These true but surprising facts are important, which is why I consider this a super fact.
Angular Momentum and Magnetic Dipoles
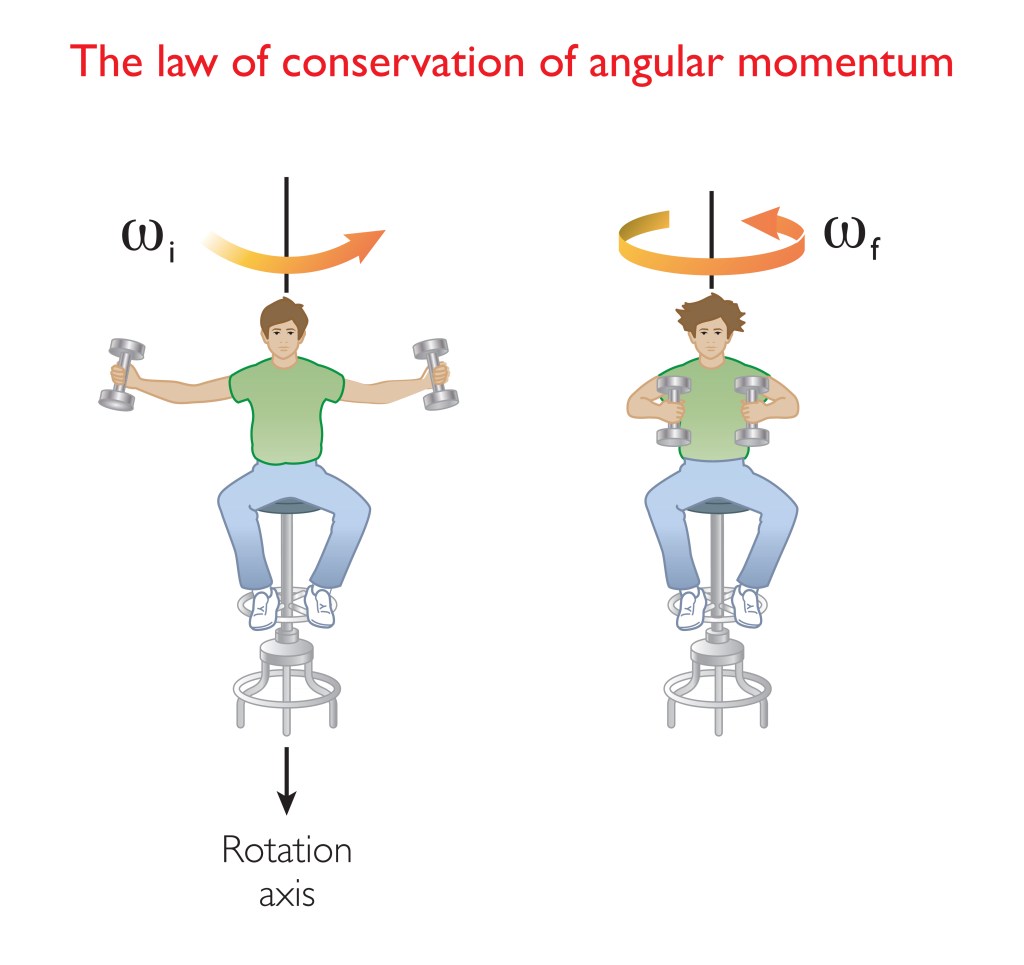
The picture above gives an example of the law of conservation of angular momentum. Bringing weights closer to the body speeds up the rotation. Another example of the law of conservation of angular momentum is when you try to tip the axis of the rotation. This would be the man tipping over. You can’t do that without applying a force. The electron, and other subatomic particles with spin, display this gyroscopic effect, like a spinning top. As mentioned, charged particles such as the electron, positron or proton, also generate a magnetic field as if they were spinning. However, the strange fact is that despite that electrons and other particles with spin display these spin properties, they aren’t spinning.
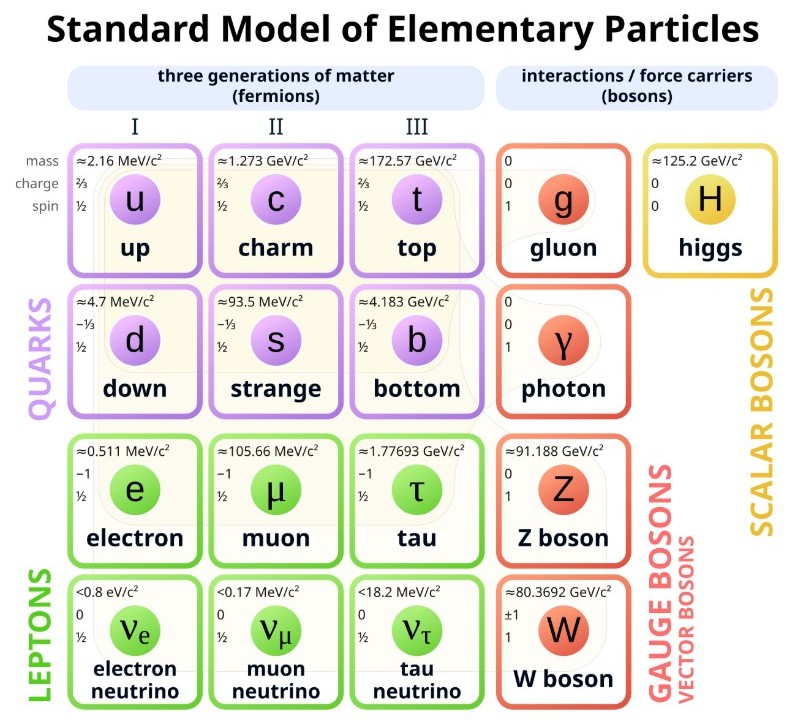
Below is an overview of the elementary particles. An elementary particle is a fundamental subatomic building block of the universe that cannot be divided into smaller components. Notice that the proton and the neutron are not listed because they are not fundamental particles. They consist of three quarks. The top number is the mass (0.511 MeV/c2 for an electron). The number below that is the charge (-1 for an electron) and the bottom number is spin (1/2 for an electron). The diagram shows three intrinsic properties per particle.
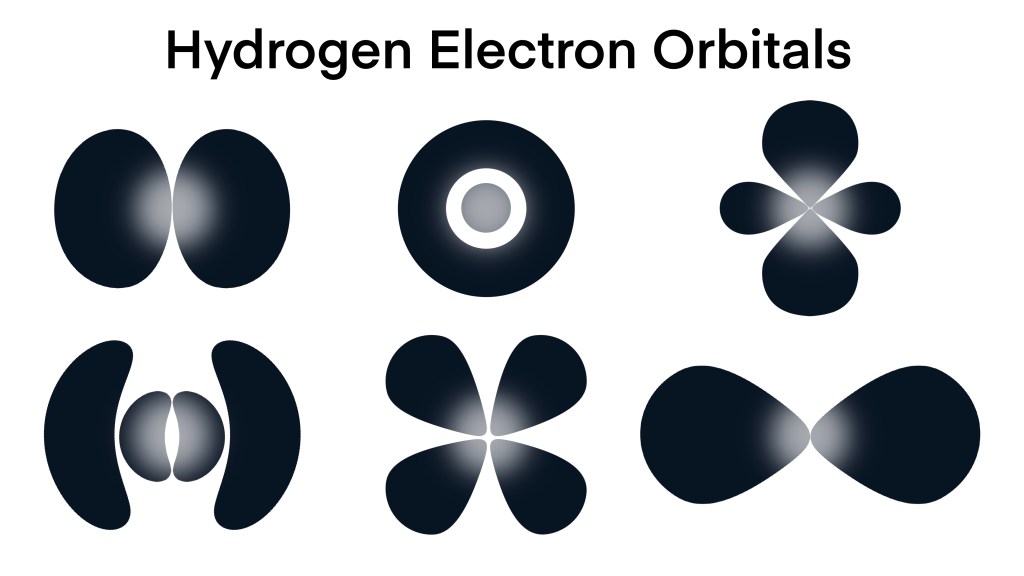
Electron Orbitals
I should explain something about particles and waves. Subatomic particles are associated with quantum waves. This is quantum mechanics. For example, an electron is in a sense both a particle and a wave, or more correctly neither. However, it exhibits both point like particle characteristics and wave characteristics depending on circumstances. Below is a picture showing the standing quantum waves representing an electron in different orbitals (different states) in a Hydrogen atom.
Hydrogen atoms only have one electron, but that electron can exist in different orbitals (sort of different orbits). A standing wave is a wave that is not spreading out, like the waves going back and forth in a bathtub. You can’t really say that the electron is orbiting the nucleus like a planet. The standing quantum wave, or electron cloud, or orbital are more accurate ways to view it.
There is some confusion as to what the standing quantum waves represent. The Copenhagen interpretation says that the electron exists in superposition, or all possible states, until measured. The quantum wave indicates the probability that you will find the electron in a certain place when you measure. The square of the amplitude of the wave is the probability that you will find the electron at that point. When you measure it and find out where the electron is the wave will collapse.
Other interpretations say that the electron is in a specific place, you just don’t know where, but again the square of the amplitude of the wave is the probability that you will find the electron at that point. Yet other interpretations say that the quantum wave is a real physical thing that guides the electron (pilot waves). The so called many-worlds-interpretation say that all possible outcomes of a measurement happens but in an infinite number of parallel universes (multi-verses).
Then some people say that the quantum wave does not exist at all, other than as a probability distribution. It represents what the observer knows about the system, nothing else. If you don’t know where the electron is, then the wave is all over the place. If you measure where it is then the wave collapses. Then we also have the you-don’t-know-what-you-are-talking-about-just-shut-up-and-calculate interpretation. The latter interpretation is focused on using the equations, for example the Schrödinger and Dirac equations, to make predictions and measurements and it ignores what’s behind the scenes. This interpretation is popular in college physics classes.
Whichever interpretation you prefer, the you-must-fully-rotate-the-wave-function-twice-to-get-back-to-start property leads to the Pauli exclusion principle.
The Pauli Exclusion Principle
The Pauli exclusion principle states that two identical fermions (such as electrons) cannot occupy the same quantum state simultaneously. For example, two electrons in the same orbital must have opposite spin +1/2 and -1/2 and you could never add a third electron. This adds structure to the atom and to the nucleus. If you did not have the Pauli exclusion principle everything could just fall into one point, and you could walk through walls. Matter as we know it could not exist. This is why fermions (electrons, muons, positrons, quarks, protons, neutrons, etc.) often are referred to as matter particles and bosons (photons, gluons, etc.) are referred to as radiation.
It turns out that that the Pauli exclusion principle is a direct result of the half spin of fermions, in other words, that you have to rotate the associated wave twice around to get back to the original.
Do you think this is confusing ? Don’t feel bad. Richard Feyman one of the most prominent pioneers of quantum mechanics and Nobel Prize winner in physics said, “I think I can safely say that nobody understands quantum mechanics”.
Other Super Facts Related to Modern Physics
- The Speed of Light In Vacuum Is a Universal Constant.
- Two events may be simultaneous for some but not for others.
- The Bizarre Reality of Black Holes.
- Every Symmetry is Associated with a Conservation Law.
- There is strong evidence for the Big Bang.
- Time Dilation Goes Both Ways.
- The Pole-Barn Paradox and Solution.
- Time is a Fourth Dimension.
- Electric Charge is not the only type of Fundamental Charge.
- Superfluid Liquids are Like Magic.
- The Edge of the Observable Universe is 46.5 billion Light Years Away.

A very interesting post, Thomas. Thank you.
LikeLiked by 1 person
Thank you so much Lynette
LikeLiked by 1 person
Thank you for this excellent paper. You take such great care to anticipate the reader’s questions and share pertinent details. I had not heard of the Pauli Exclusion Principle before, nor of motion as a “conversation.” Brilliant writing, Thomas. Thank you!
LikeLiked by 1 person
Thank you so much for your kind words Suzette. About “conversation.”, that’s auto correct in word. It was supposed to be “conservation.” of angular momentum like a spinning top resisting to tip over unless you apply a force. The electron behaves the same way but without rotating.
LikeLiked by 1 person
Thank you for the great clarification, Thomas. I always learn new things from your posts. Much appreciated!
LikeLiked by 1 person
You are so very kind Suzette. I appreciate your support.
LikeLike
The diagram showing the law of conversation of angular momentum is very intriguing. I had no idea that electron’s aren’t actually spinning; that is indeed an excellent and little-known fact!
LikeLiked by 1 person
Thank you so much Ada. About “conversation.”, that’s auto correct in word. It was supposed to be “conservation.” of angular momentum like a spinning top resisting to tip over unless you apply a force. The electron behaves the same way but without spinning. It is very confusing how it can have the properties of spinning (spinning a lot too) even though it does not actually spin.
LikeLike
Oh wow… amazing!
LikeLiked by 1 person
Thank you Chris
LikeLiked by 1 person
The angular momentum example reminds me of the spins that figure skaters put at the end of their routine, when they start the spin with a leg and their arms out and then pull them in close to make the spin faster.
LikeLiked by 1 person
Yes exactly, or like a spinning top, or a gyroscope, plus the magnetic field that would be created from a charge spinning. The electron have all those characteristics, but when all is said and done, it still does not spin, not physically. That is a mystery.
LikeLiked by 1 person
Someday, someone will figure it out. Maybe you?
LikeLiked by 1 person
Thank you for the confidence Anneli. However, I started out in physics but switched to engineering, and now I am old.
LikeLiked by 1 person
This is one of the post where I understand and appreciate the overarching message but the details lose me a bit.
LikeLiked by 1 person
Sorry, it is a bit abstract and I might have said more than I should. Basically, the electron act like a spinning top, and generates a magnetic field, like a magnetic dipole, like spinning charged objects do. However, it isn’t spinning. It is intrinsic.
Secondly, you have to rotate the whole system (particle/wave/orbital) twice around to get back to where you started, which is strange. This strange feature also makes it so fermions, things with half spin, (electrons, positrons, protons, …) can’t be in the same place/state. This keeps them separated and gives structure to atoms and everything else that fermions are part of. You can’t put everything in a little dot and you can’t walk through walls.
LikeLike
Thanks, this makes perfect sense. Good information.
LikeLiked by 1 person
Thank you so much Robbie
LikeLike
I thought it was supposed to be conservation, I see your other comments on the spell-check error. I think you describe this better than my university profs ever could. Well done. Maggie
LikeLiked by 1 person
Thank you so much for your very kind words Maggie
LikeLike
Pretty cool. I learned something new and some of it over my head 🙂 The top spinning and not tipping over was pretty cool. Always wondered about that.
LikeLiked by 1 person
Yes the top spinning and not tipping over unless you apply a force is an example of the conservation of angular momentum and an electron have that feature without actually spinning. Thank you Kevin.
LikeLike
Thanks for another good physics post. Despite my background in physics, I didn’t realize that electrons do not actually spin. That said, I have tried the angular momentum experiment you illustrate where you sit or stand on a spinning surface and hold weights, then hold the weights away from you and then bring them in close to see how thing change.
LikeLike